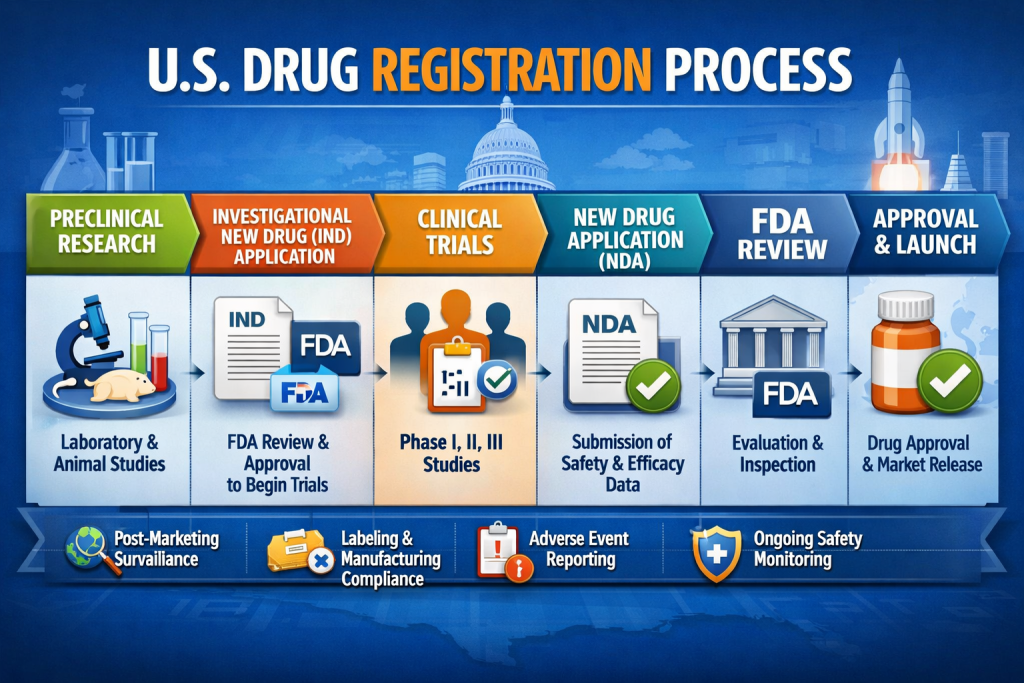
The United States pharmaceutical market is one of the most tightly regulated in the world. Any company planning to manufacture, export, or distribute drug products in the US must comply with US Drug Registration requirements enforced by the U.S. Food and Drug Administration (FDA). Without proper registration and listing, drugs cannot be legally sold or imported into the United States.
This article explains the FDA drug registration framework, regulatory obligations, and why professional compliance support is critical for uninterrupted market access.
Understanding US Drug Registration
US Drug Registration is a regulatory obligation under the Federal Food, Drug, and Cosmetic Act (FD&C Act). It applies to all entities involved in drug manufacturing or processing for the US market.
The process consists of two mandatory components:
- FDA Drug Establishment Registration
- FDA Drug Product Listing
Both steps are required regardless of whether the company is located inside or outside the United States.
Importantly, FDA registration is not a product approval. Registration only confirms that the FDA has been notified about the facility and the drug products being marketed.
Who Is Required to Register?
FDA drug registration applies broadly across the pharmaceutical supply chain.
Entities Covered
- Prescription drug manufacturers
- OTC drug manufacturers
- Contract manufacturing organizations
- Repackers and relabelers
- Foreign manufacturers exporting drugs to the US
Foreign companies must also designate a US Agent, who acts as the FDA’s official contact for compliance communication and inspections.
FDA Drug Establishment Registration
Every physical site involved in drug manufacturing or processing must be registered individually with the FDA.
Key Registration Rules
- Registration is mandatory and annual
- Renewal window: October 1 to December 31
- Applies to both US and non-US facilities
- Required before commercial distribution or import
Failure to renew establishment registration can lead to inactive status, import refusals, and regulatory enforcement actions.
FDA Drug Listing Requirements
Once the establishment is registered, each drug product must be listed with the FDA.
Drug listing submissions include detailed product information such as:
- Drug name (brand and generic)
- Dosage form and strength
- Route of administration
- Labeler information
- National Drug Code (NDC)
- Product labeling details
All listings must be submitted electronically using the FDA’s Structured Product Labeling (SPL) system.
National Drug Code (NDC): Why It Matters
The National Drug Code (NDC) is a unique identifier assigned to drugs listed with the FDA. While the FDA does not approve NDC numbers, they are essential for:
- US commercial distribution
- Pharmacy and hospital systems
- Insurance and reimbursement processes
- Import and export documentation
Incorrect NDC configuration is a common reason for FDA listing rejection or data correction requests.
FDA Registration for Foreign Drug Manufacturers
Foreign manufacturers face additional regulatory responsibilities, including:
- Appointment and maintenance of a US Agent
- Coordination with FDA during inspections
- Compliance with US import regulations
- Consistency between labeling, listing, and shipment data
Any discrepancy can result in shipment delays, port detentions, or import alerts.
Common Compliance Risks
Companies often encounter FDA registration issues due to:
- Incorrect or incomplete SPL submissions
- Labeling mismatches
- Missing annual renewals
- Improper US Agent designation
- Misunderstanding the difference between registration and approval
Even minor errors can delay product launches or trigger regulatory scrutiny.
Role of FDA Drug Registration Consultants
Managing FDA drug registration requires regulatory expertise, technical knowledge, and continuous monitoring. Errors can disrupt supply chains and harm commercial credibility.
XPRO America, a US FDA Consultancy, supports pharmaceutical companies through the complete FDA drug registration lifecycle. Services include establishment registration, drug listing, SPL preparation, NDC setup, US Agent representation, and annual compliance maintenance.
Organizations seeking structured and compliant FDA registration support may reach the consulting team via support@xproamerica.com, where regulatory specialists provide tailored guidance based on product type and market strategy.
Conclusion
US Drug Registration is a critical regulatory foundation for accessing the American pharmaceutical market. While the process is procedural, it demands precision, regulatory awareness, and ongoing updates.
By maintaining accurate FDA establishment registration and drug listings—and working with an experienced US FDA consultancy—companies can reduce regulatory risk, avoid costly delays, and ensure long-term compliance in the US market.
Leave a Reply